  
One hot summer day they climbed the huge hill next to our house to dig a hole to hide their bottle-cap collection. I was able to use water density once to at least play a trick on them, though. Growing up with an older brother was difficult, especially when he had his friends over, for their favorite activity was thinking of ways to antagonize me. The rounded value of 1 g/ml is what you'll most often see, though. A common unit of measurement for water's density is gram per milliliter (1 g/ml) or 1 gram per cubic centimeter (1 g/cm 3).Īctually, the exact density of water is not really 1 g/ml, but rather a bit less (very, very little less), at 0.9998395 g/ml at 4.0° Celsius (39.2° Fahrenheit). Density is just the weight for a chosen amount (volume) of the material. As long as an object is made up of molecules, and thus has size or mass, it has a density. The definition of density, makes a lot more sense with a little bit of explanation. If you're not still in school, then you probably forgot you ever even heard it. On Earth, you can assume mass is the same as weight, if that makes it easier. So, one molecule of H 2O weighs 2.9915 × 10 -23 grams.If you're still in school, you've probably heard this statement in your science class: " Density is the mass per unit volume of a substance". Using this constant and the molar mass above, the formula to find the weight of one H 2O molecule is: That’s a pretty large number, so it’s typically represented using scientific notation. Therefore, the molar mass of H 2O is 18.01528 g/mol, or 18.01528 grams per mole.Īfter finding the molar mass, you can find the weight of a molecule using Avogadro’s constant, which states that one mole is equal to 6.02214076 × 10 23 elementary units of matter, such as a molecule. Since water contains two hydrogen atoms and one oxygen atom, the molar mass formula for H 2O is: The mass of hydrogen is 1.00794 g/mol, and oxygen is 15.9994 g/mol. You can find the molar mass of H 2O by finding the atomic mass of each element in the molecule and adding them together. 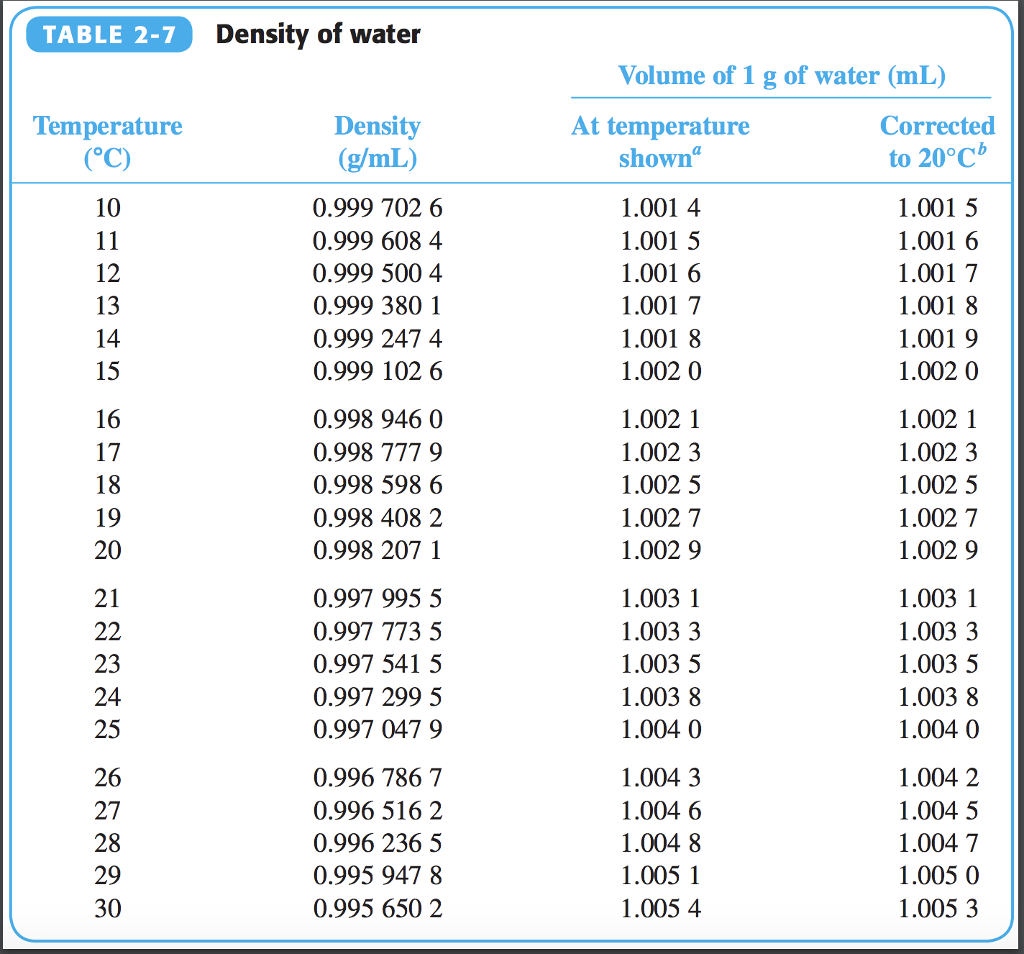
To determine the weight of an H 2O molecule, you need to first find its molar mass. 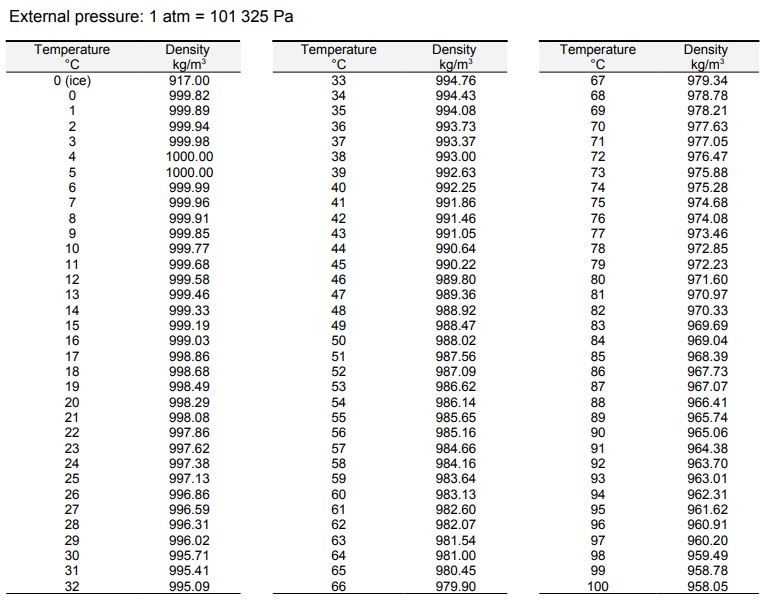
It’s an oxygen hydride that consists of a single oxygen atom bonded to two hydrogen atoms. In chemistry, water is also referred to as H 2O. How Much Does One Molecule of H 2O Weigh? Working on a plumbing project? Use our pipe volume calculator to calculate the volume and weight of the water in your plumbing system. You can use the calculator or weight chart above for the full five-gallon weight range. Trying to figure out how much a 5-gallon bucket of water weighs? At room temperature, five gallons of water weighs 41.64 pounds, but at 32.2 ☏, it weighs 41.727 pounds. Table showing the weight of one US gallon of water at different temperatures Temperature ( ☏ / ☌ ) The table below shows the weight at various temperatures. So, just how much does a gallon of water weigh? One gallon of water weighs between 8.288489 and 8.345404 pounds (lb), depending on the temperature. But, since the density of water fluctuates with the temperature, the weight of a gallon of water also changes slightly. We mentioned above that one gallon of water weighs 8.345 pounds at 39.2 ☏ when the density of water is exactly equal to 1.0 g/cm³. Table showing the density of water at different temperatures. The chart below shows the density of water at various temperatures, according to the US Department of the Interior. Thus, the warmer the water is, the more volume it will consume and the lower its density will be. As the energy in the molecules rises, the particles within move and vibrate more, causing them to take up more space. Īt a molecular level, as the temperature rises, the heat of the water molecules increases, which increases their energy. As the temperature of water rises, it expands, causing it to increase in volume slightly. The Temperature Affects the Weight of WaterĪs we mentioned above, temperature affects the density of water, and thus its precise weight will vary with the temperature. Table showing the weight of water in various units of measure. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
